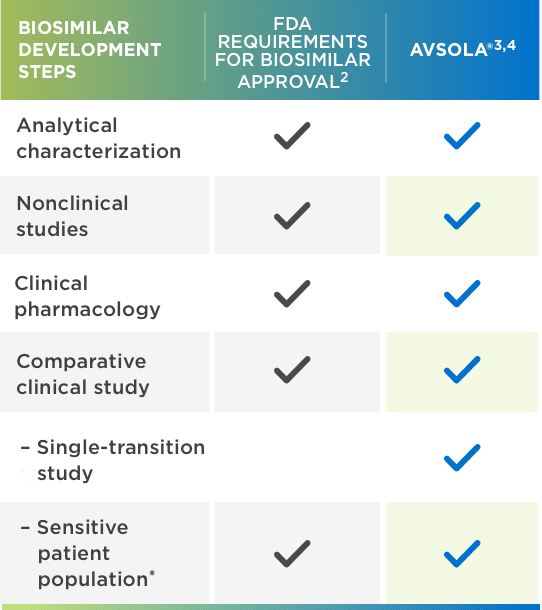
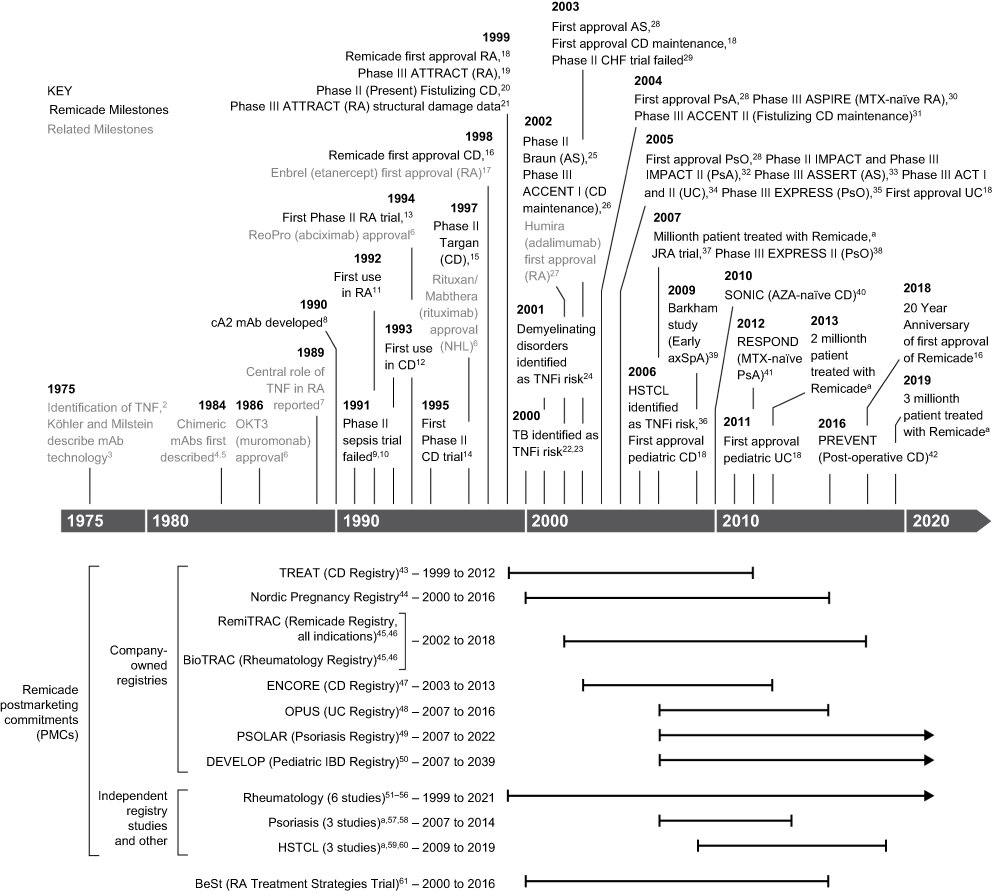
Biosimilar strategy for infliximab by MedDrive™ includes the reference drug - Prime Therapeutics LLC

Biosimilar medicine fact sheet – infliximab | Australian Government Department of Health and Aged Care
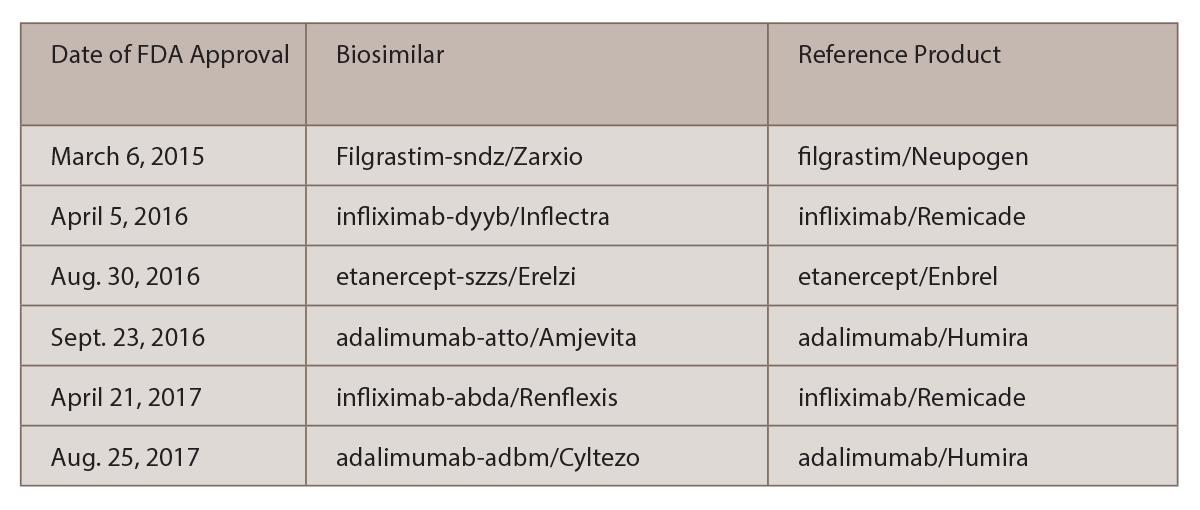
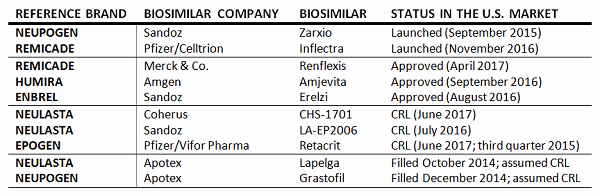
Report on EU's Experience with Biosimilar Drugs Released: Will U.S. Experience Be Similar? - Page 2 of 5 - The Rheumatologist