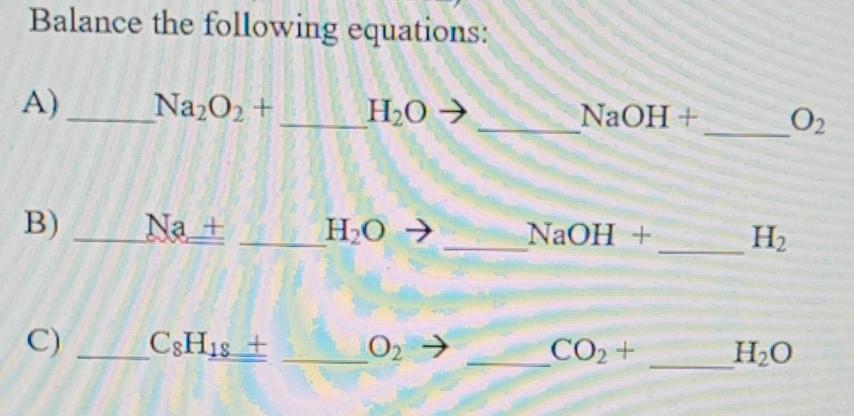
Q:12 Balance the following chemical reactions- 17a) CO +H2 → CH3OH b) Na + H20 → NaOH + H2 C) BaCl2 + Alz(SQ)3 → BaSO4 + AlCl3

Balancing Chemical Equations Worksheet Answers Check these answers against the work you did on the six problems. - ppt download

Name the reactions and products of the following. Na+ H2O NaOH+ H2. Also balance the unit. - Brainly.in

Oxidation Number method Na+H2O=NaOH+H2 Balance the chemical equation by oxidation Number method. - YouTube
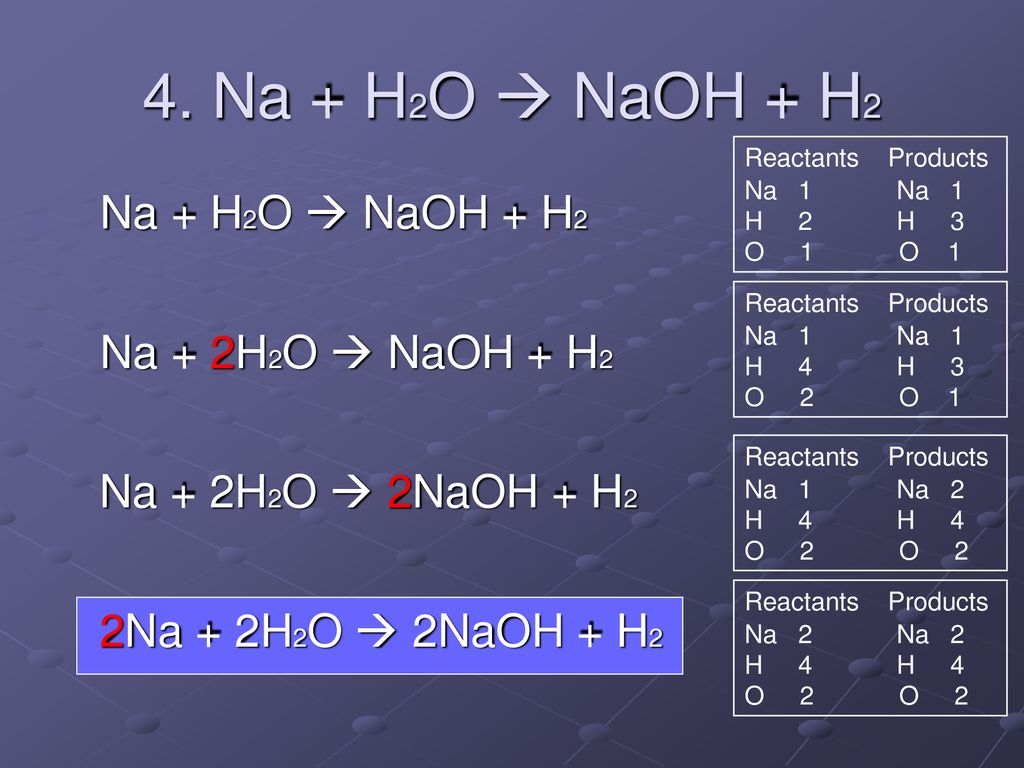
Chapter 6 – 3 Warm - Up 1. What are the 6 different types of chemical reactions we talked about in class? 2. What type of reaction is: 2 Na + Cl2 ppt download















![ANSWERED] The following chemical equation, 2 Na(s)... - Inorganic Chemistry - Kunduz ANSWERED] The following chemical equation, 2 Na(s)... - Inorganic Chemistry - Kunduz](https://media.kunduz.com/media/sug-question/raw/50091904-1658915707.4594715.jpeg)